Ions
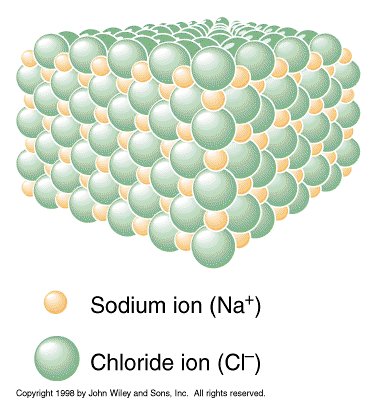
If an atom has unequal numbers of protons and electrons, it will be charged and is called an ion. The reaction below shows how ions form.

The tendency is to gain or lose electrons until the number of electrons remaining equals that of the nearest noble gas.
Ionic compounds (salts) are held together by the electrical attraction between oppositely charged ions. This is called ionic bonding.